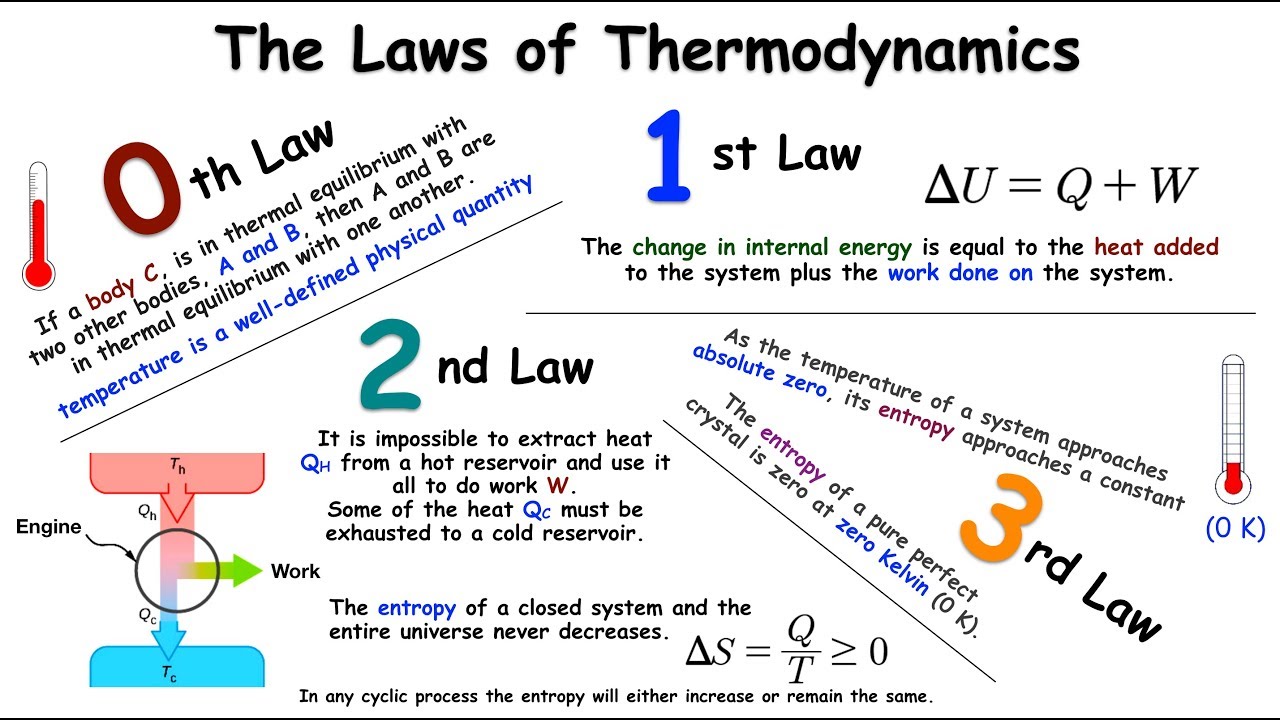 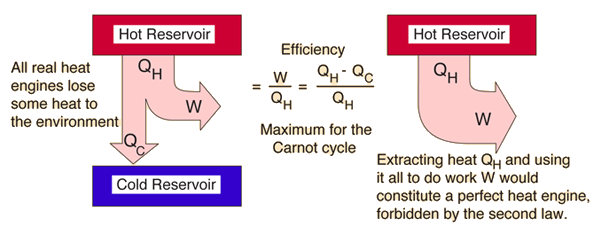
The first law of thermodynamics states that energy can neither be created nor be destroyed. 
Therefore we can say that, there is a net increase in the entropy. The entropy increased in this process is much greater than the entropy decreased. The ice cube then absorbs the same amount of heat which was released by water and therefore its entropy increases. 
The water releases off heat and the ice cube melts. When we put an ice cube in a cup with water at room temperature. Real life Example of second law of thermodynamics is that: Thermodynamics has three laws and the second law of thermodynamics states that the entropy of a system never decreases and it is always positive.Įntropy refers to the measure of the molecular disorder or we can say the randomness of a system. Thermodynamics have three laws out of which the second law of thermodynamics states that the net entropy of a system always increases and never decreases.Īs we know, thermodynamics is a branch of science which deals with work done, heat, internal energy on a system by surroundings or on surroundings by the system. Hint :Thermodynamics is a branch of science which deals with work done, heat, internal energy on a system by surroundings or on surroundings by the system.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
